 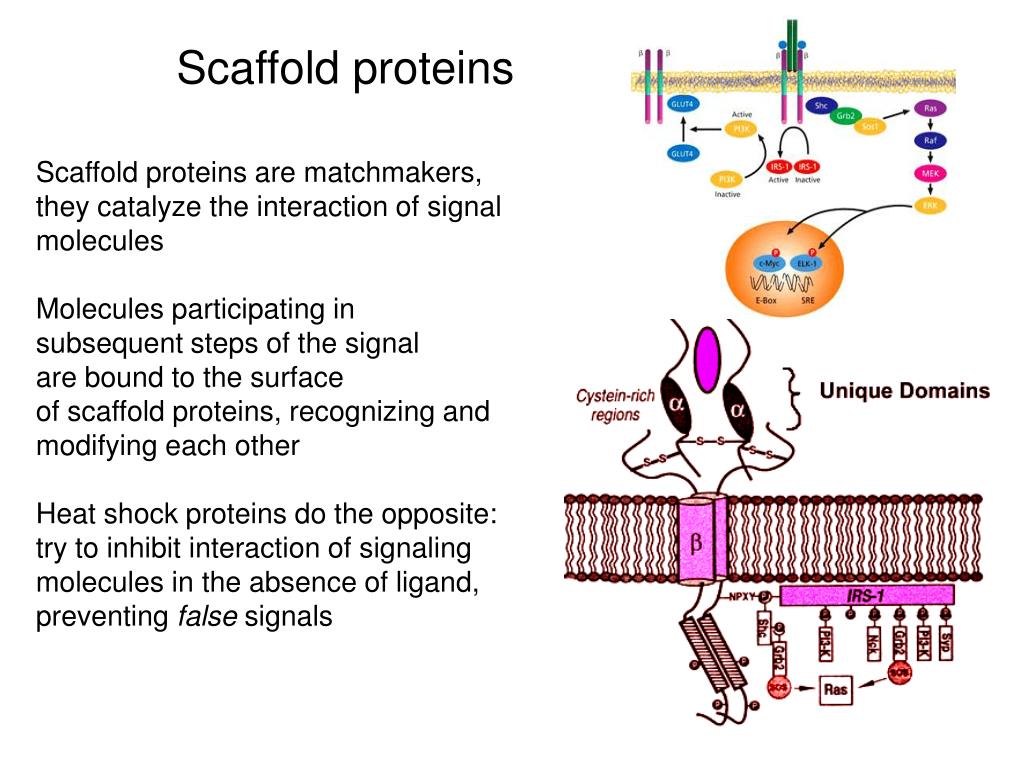
Dlg5 studies in mammals emphasized a function in epithelial morphogenesis, the knock-out mouse showing mild defects of adherens junction and epithelial polarity in the kidney, the lung and the brain. A more divergent member of the family, Dlg5, is also found in fly and mammals with a conserved architecture: a coiled-coil domain, 4 PDZ domains and a MAGUK domain. Four paralogs of fly Dlg, Dlg1 to Dlg4, are found in mammals. ĭiscs Large (Dlg) is a MAGUK protein that was identified in Drosophila for its function in epithelial polarity as a determinant of the lateral domain and the neoplastic effect of its mutation. MAGUK proteins also emerge as important modulators of phase separation in cells. Some MAGUK domains recognize phosphopeptides, whereas some others work in cooperation with the adjacent PDZ domain, reinforcing the affinity of the latter for a specific partner. These domains are usually flanked by one or several PDZ domains and potentially other protein-protein interaction domains. MAGUK domain is a structural unit formed by SH3 domain next to a non-catalytically active guanylate kinase domain. Membrane-associated guanylate kinase (MAGUK) proteins are typical examples of scaffold proteins. They allow the formation of complexes that are determinant, for instance, for cell polarity, cell adhesion or that are used as a platform for various signaling events. These proteins are devoid of catalytic activity but contain multiple domains of protein-protein interaction. Among the protein classes involved in such processes, many of them are scaffold proteins. The accurate development of an organ or an organism requires a robust coordination size and shape control, both at the cell and tissue scales. Moreover, they reveal a differential regulation of E-cadherin and N-cadherin apical localization. Together our data show that Dlg5 own several conserved functions that are independent of each other in regulating growth, cell polarity and cell adhesion. Genetic analyses indicate that junction and polarity defects are independent. Dlg5 is also essential, in the same cells, for the presence at Adherens junctions of N-Cadherin, but not E-Cadherin. 
Moreover, epithelia cells mutant for Dlg5 also show a reduction of apical domain determinants, though not sufficient to induce a complete loss of cell polarity. However, other observations indicate that Dlg5 regulates growth by at least another way that may involve Myc protein but nor PI3K neither TOR pathways. Our results are consistent with Dlg5 modulating hippo pathway in the wing disc, including the impact on cell size, a defect that is reproduced by the loss of yorkie. Dlg5 is required for growth at a cell autonomous level in several tissues and at the organism level, affecting cell size and proliferation. Here, we generated null mutants for Drosophila Dlg5 and show that it owns similar functions in growth and epithelial architecture. Mutant mouse for the MAGUK protein Discs large 5 (Dlg5) presents a general growth impairment and moderate morphogenetic defects. Scaffold proteins support a variety of key processes during animal development. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
